Chemical and Process Engineering Resources

One of the most popular methods of moving solids in the chemical industry is pneumatic conveying. Pneumatic conveying refers to the moving of solids suspended in or forced by a gas stream through horizontal and/or vertical pipes. Pneumatic conveying can be used for particles ranging from fine powders to pellets and bulk densities of 16 to 3200 kg/m3 (1 to 200 lb/ft3).
Dilute Phase versus Dense Phase Conveying
The specifying engineer typically has four choice in specifying a pneumatic conveying system.
- Dilute phase vacuum operation
- Dilute phase pressure operation
- Dilute phase pressure-vacuum operation
- Dense phase pressure operation
Vacuum systems allow multiple product inlets through the use of simple diverter valves. However, it becomes costly to have multiple destinations because each must have its own filter receiver with partial vacuum capability. Vacuum systems are also more "distance sensitive" than pressure systems due to the maximum pressure differential of 5.5 to 6.0 psi. Dilute phase pressure systems can easily achieve pressure differentials of 12 psi. Pressure-vacuum operation (utilizing both methods) are sometimes ideal for a given conveying setup. A very common application is the unloading of a standard railcar. Since the cars cannot be pressurized, air is pulled from the outside, through the car (carrying solids with it) to a filter. Then after the filter, a blower can be used to forward the solids to the final receiver.
The choice between dilute and dense phase operation is typically dependent on the solids properties. For example, the lower velocity bulk phase operation is popular ofr highly abrasive products or for those that degrade easily. For example, this method is popular in transporting kaolin clay.
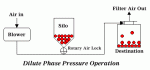 | Â | 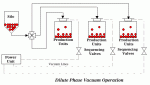 | Â | 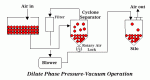 |
| Figure 1: Dilute Phase Pressure Conveying | Â | Figure 2: Dilute Phase Vacuum Conveying | Â | Figure 3: Dilute Phase Pressure-Vacuum Conveying |
Designing a System for Dilute Phase Operation
Considering designing a pneumatic conveying system yourself? Probably not a good idea. There's as much art involved as there is science and such a design should be left to professionals. Consider that even different grades of the same material have been known to convey differently. Testing is a must (as you'll see from the method below). Before you can even make any good judgements from the method presented here, you need to know solid friction factor for your solids (which we'll discuss later) and the minimum gas velocity required to move your particles. So, if you're involved in designing a system from the ground up, seek assistance from reputatable people in the field of conveying. If you're already familiar with your solids, the method below can be used to examine the pressure loss expected in your system. The method presented here is very good and has been stood the test of real systems over time.
The design method presented here is based on the work of Dr. F.A. Zenz and Dr. D.F. Othmer as published in their book "Fluidized and Fluid Particle Systems" published in 1960 (see References). This method was presented by A. T. Agarwal of Pneumatic Conveying Consulting Services LLC in the January/February 2005 issue of Powder Handling and Processing. This method has been widely used and is generally found to be within 10% of measured pressure losses.
Pressure losses experienced in pneumatic conveying systems are the result of the following forces:
Friction of the gas on the inside of the pipe, force required to move the solids through the pipe, forces required to support the weight of the solid and the gases in vertical pipe runs, force required to accelerate the solids, and friction between the solids and the inside of the pipe.
Friction losses as the result of the solids being in contact with the inside of the pipe are usually very small and can be neglected when considering dilute phase transport.
Please be aware that using air as a carrier gas should be investigated thoroughly. When some powders are mixed with oxygen, they form an explosive mixture!

 FB
FB

0 Comments