Chemical and Process Engineering Resources

Equipment Used in Crystallization
Tank Crystallizers
This is probably the oldest and most basic method of crystallization. In fact, the "pot of salt water" is a good example of tank crystallization. Hot, saturated solutions are allowed to cool in open tanks.Â
Scraped-Surface Crystallizers
An example may be the Swenson-Walker crystallizer consisting of a trough about 2 feet wide with a semi-circular bottom. The outside is jacketed with cooling coils and an agitator blade gently passes close to the trough wall removing crystals that grow on the vessel wall.
Forced Circulating Liquid Evaporator-Crystallizer
Just as the name implies, these crystallizers combine crystallization and evaporation, thus the driving forces toward supersaturation. The circulating liquid is forced through the tubeside of a steam heater. The heated liquid flows into the vapor space of the crystallization vessel. Here, flash evaporation occurs, reducing the amount of solvent in the solution (increasing solute concentration), thus driving the mother liquor towards supersaturation. The supersaturated liquor flows down through a tube, then up through a fluidized area of crystals and liquor where crystallization takes place via secondary nucleation. Larger product crystals are withdrawn while the liquor is recycled, mixed with the feed, and reheated.
 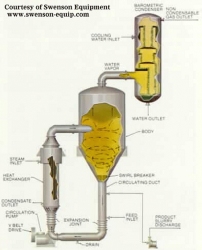 |
| Figure 4: Forced Circulating Liquid Evaporator-Crystallizer |
Circulating Magma Vacuum Crystallizer
In this type of crystallizer, the crystal/solution mixture (magma) is circulated out of the vessel body. The magma is heated gently and mixed back into the vessel. A vacuum in the vapor space causes boiling at the surface of the liquid. The evaporation causes crystallization and the crystals are drawn off near the bottom of the vessel body.
 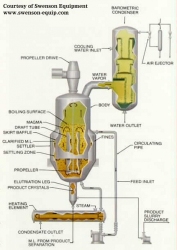 |
| Figure 5: Circulating Magma Vacuum Crystallizer |
References
- Price, Chris J., "Take Some Solid Steps to Improve Crystallization", Chemical Engineering Progress, September 1997, p. 34.
- Geankoplis, Christie J., Transport Processes and Unit Operations, 3rd Ed., Prentice Hall, New Jersey, 1993, ISBN: 0-13-930439-8
- Brown, Theodore L., Chemistry: The Central Science, 5th Ed., Prentice Hall, New Jersey, 1991, ISBN: 0-13-126202-5
- Swenson Process Equipment web site at http://www.swensontechnology.com/
Â
Â

 FB
FB


0 Comments