Chemical and Process Engineering Resources
Experienced Based Rules of Chemical Engineering
Nov 08 2010 01:00 PM | Chris Haslego
Experience is typically what turns a good engineer into a great engineer. An engineer that can look at a pipe and a flowmeter and guess the pressure drop within 5%. Someone who can at least estimate the size of a vessel without doing any calculations.
When I think of such rules, two authors come to my mind, Walas and Branan. Dr. Walas' book, Chemical Process Equipment: Selection and Design has been widely used in the process industry and in chemical engineering education for years. Mr. Branan has either helped write or edit numerous books concerning this topic. Perhaps his most popular is Rules of Thumb for Chemical Engineers. Here, I'll share some of these rules with you along with some of my own. Now, be aware that these rules are for estimation and are not necessary meant to replace rigorous calculations when such calculations should be performed. But at many stages of analysis and design, these rules can save you hours and hours.
Download these rules of thumb in MS Excel format.
Physical Properties
| Property | Units | Water | Organic Liquids | Steam | Air | Organic Vapors |
| Â | Â | Â | Â | Â | Â | Â |
| Heat Capacity | KJ/kg 0C | 4.2 | 1.0-2.5 | 2.0 | 1.0 | 2.0-4.0 |
| Â | Btu/lb 0F | 1.0 | 0.239-0.598 | 0.479 | 0.239 | 0.479-0.958 |
| Density | kg/m3 | 1000 | 700-1500 | Â | 1.29@STP | Â |
| Â | lb/ft3 | 62.29 | 43.6-94.4 | Â | 0.08@STP | Â |
| Latent Heat | KJ/kg | 1200-2100 | 200-1000 | Â | Â | Â |
| Â | Btu/lb | 516-903 | 86-430 | Â | Â | Â |
| Thermal Cond. | W/m 0C | 0.55-0.70 | 0.10-0.20 | 0.025-0.070 | 0.025-0.05 | 0.02-0.06 |
| Â | Btu/h ft 0F | 0.32-0.40 | 0.057-0.116 | 0.0144-0.040 | 0.014-0.029 | 0.116-0.35 |
| Viscosity | cP | 1.8 @ 0 0C | **See Below | 0.01-0.03 | 0.02-0.05 | 0.01-0.03 |
| Â | Â | 0.57 @ 50 0C | Â | Â | Â | Â |
| Â | Â | 0.28 @ 100 0C | Â | Â | Â | Â |
| Â | Â | 0.14 @ 200 0C | Â | Â | Â | Â |
| Prandtl Number | Â | 1-15 | 10-1000 | 1.0 | 0.7 | 0.7-0.8 |
** Viscosities of organic liquids vary widely with temperature
Liquid densities vary with temperature to this approximation:
| Eq. (1) |
Gas densities can be calculated by:
| Eq. (2) |
The boiling point of water can be approximated as a function of pressure by:
| Eq. (3) |
Â
Materials of Construction
| Material | Advantage | Â | Disadvantage |
| Carbon Steel | Low cost, easy to fabricate, abundant, most common material. Resists most alkaline environments well. | Â | Very poor resistance to acids and stronger alkaline streams. More brittle than other materials, especially at low temperatures. |
| Stainless Steel | Relatively low cost, still easy to fabricate. Resist a wider variety of environments than carbon steel. Available is many different types. | Â | No resistance to chlorides, and resistance decreases significantly at higher temperatures. |
| 254 SMO (Avesta) | Moderate cost, still easy to fabricate. Resistance is better over a wider range of concentrations and temperatures compared to stainless steel. | Â | Little resistance to chlorides, and resistance at higher temperatures could be improved. |
| Titanium | Very good resistance to chlorides (widely used in seawater applications). Strength allows it to be fabricated at smaller thicknesses. | Â | While the material is moderately expensive, fabrication is difficult. Much of cost will be in welding labor. |
| Pd stabilized Titanium | Superior resistance to chlorides, even at higher temperatures. Is often used on sea water application where Titanium's resistance may not be acceptable. | Â | Very expensive material and fabrication is again difficult and expensive. |
| Nickel | Very good resistance to high temperature caustic streams. | Â | Moderate to high expense. Difficult to weld. |
| Hastelloy Alloy | Very wide range to choose from. Some have been specifically developed for acid services where other materials have failed. | Â | Fairly expensive alloys. Their use must be justified. Most are easy to weld. |
| Graphite | One of the few materials capable of withstanding weak HCl streams. | Â | Brittle, very expensive, and very difficult to fabricate. Some stream components have been know to diffusion through some types of graphites. |
| Tantalum | Superior resistance to very harsh services where no other material is acceptable. | Â | Extremely expensive, must be absolutely necessary. |
Compressors and Vacuum Equipment
A. The following chart is used to determine what type of compressor is to be used:
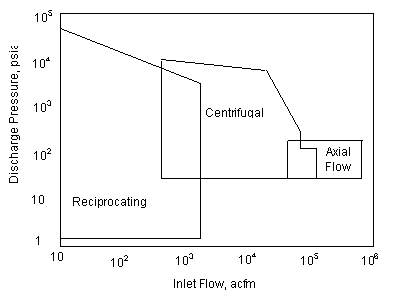 |
| Figure 1: Range Chart for Various Types of Compressors |
B. Fans should be used to raise pressure about 3% (12 in water), blowers to raise to less than 2.75 barg (40 psig), and compressors to higher pressures.
C. The theoretical reversible adiabatic power is estimated by:
| Power = m z1 R T1 [({P2 / P2}a - 1)] / a | Eq. (4) |
where:
T1 is the inlet temperature
R is the gas constant
z1 is the compressibility
m is the molar flow rate
a = (k-1)/k
k = Cp/Cv
D. The outlet for the adiabatic reversible flow, T2 = T1 (P2 / P1)a
E. Exit temperatures should not exceed 204 °C (400 °F).
F. For diatomic gases (Cp/Cv = 1.4) this corresponds to a compression ratio of about 4
G. Compression ratios should be about the same in each stage for a multistage unit, the ratio = (Pn / P1) 1/n, with n stages.
H. Efficiencies for reciprocating compressors are as follows:
65% at compression ratios of 1.5
75% at compression ratios of 2.0
80-85% at compression ratios between 3 and 6
I. Efficiencies of large centrifugal compressors handling 2.8 to 47 m3/s (6000-100,000 acfm) at suction is about 76-78%
J. Reciprocating piston vacuum pumps are generally capable of vacuum to 1 torr absolute, rotary piston types can achieve vacuums of 0.001 torr.
K. Single stage jet ejectors are capable of vacuums to 100 torr absolute, two stage to 10 torr, three stage to 1 torr, and five stage to 0.05 torr.
L. A three stage ejector requires about 100 lb steam/lb air to maintain a pressure of 1 torr.
M. Air leakage into vacuum equipment can be approximated as follows:
Leakage = k V(2/3)
where:
k =0.20 for P >90 torr, 0.08 for 3 < P < 20 torr, and 0.025 for P < 1 torr
V = equipment volume in cubic feet
Leakage = air leakage into equipment in lb/h
Download these rules of thumb in MS Excel format.

 FB
FB

13 Comments
Progreso es compartir el conocimiento. Gracias
Thanks for the good information.
amazing. thank you!
thank u for information.
nice
design calc. of heat transfer equipment needed
good
Thanks so much
all process engineering at a glance, excellent
really, this is a very helpful data.
i need one more help.
I need the vapor pressure data of cetyl chloride, if any one can help me...please do the needful.
very nice.