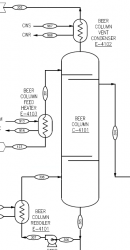
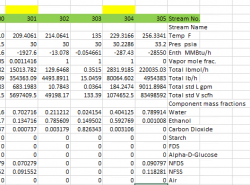
I am trying to understand a mass balance sheet for an ethanol plant. Looking at the beer column overhead vapor here's the original conditions:
flow 80,064 lb/hr
vapor mole frac. 1
mass fractions:
-water 0.44125
-ethanol 0.595875
Enthalpy -287.43 MMBtu/h
Temp 229.3166F
Press 30.2286 psia
I'm not a chemical engineer, so having a little trouble understanding the enthalpy value.
I'm using NIST Refprop software, and for that same mixture at that temperature and pressure their enthalpy value for vapor phase is 696.99 btu/lbm
multiply that by the flowrate and i'm getting 55.8MMBtu/h.
I'm sure I'm missing some fundamental principle, but i'm stumped as to why it's a negative value and so much more than what i'm coming up with.
Any help is greatly appreciated. Thanks!

 FB
FB