 Dear All,
Dear All,Boiler feedwater is often obtained from raw water by ion-exchange process utilizing vertical cylindrical vessels which contain ion-exchange resins of various types. These resins remove dissolved salts from the water and replace them with Hydrogen (H+) and hydroxyl (OH-) thus producing pure, mineral free boiler feed quality water.
Resins which replace postively charged ions such as Calcium (Ca++), Magnesium (Mg++), Sodium (Na+) etc. are called "cation" resins and they are regenerated with an acid (sulphuric or hydrochloric) to replace the hydrogen ions which were exchanged for the "cations". More often sulphuric acid is employed for regeneration since it is cheaper than hydrochloric acid and can be stored in carbon steel vessels at ambient temperatures. Hydrochloric acid is more efficient in regeneration than sulphuric acid but storage and handling is expensive and more safety concerns need to be addressed. HCl storage requires elastomer-lined steel vessels and may require fume exposure protection measures such as vent scrubbers due to its high volatility.
Resins which replace negatively charged ions such as Sulphate (SO4--), Chloride (Cl-), Carbonate (CO3--) etc. are called anion resins and they are regenerated with caustic (NaOH) to replace the hydroxyl ions which were exchanged by the "anions".
There are weak and strong cation as well as weak and strong anion resins. In general, the weak resins are more efficient in terms of regeneration chemicals (acid or caustic) consumption. However, these resins are also limited in their capability to remove ions. For example, cation removal with Weak Acid Cation (WAC) resin is limited to the amount of Bicarbonate (HCO3--) alakalinity in the water; and anion removal is limited to sulfides, chlorides and nitrates in the acidic form when using Weak Base Anion (WBA) resin.
Some cations and anions will slip through the ion exchange system and this is called "leakage".The amount aof leakage is a function of several factors such as:
a. Influent water quality
b. Type of resin employed for the cation and anion exchangers
c. Regeneration chemical type and dosage
There are some design guidelines / parameters for fixed bed ion exchange (excluding packed beds) units and forced draft degasifiers which are as follows:
1. Recommended influent water quality to avoid plugging, fouling and deterioration of resins is provided in the attached table:
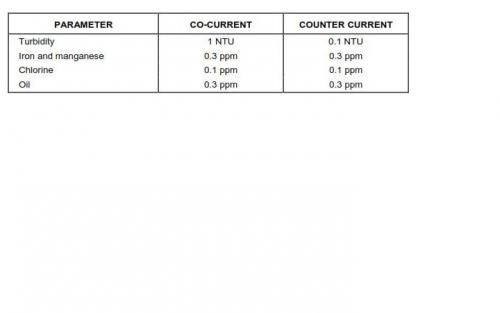
2. Flowrates for design of vessel diameter should be kept in the 2 - 15 gpm/ft2 (18 gpm/ft2 if uniform resin is used) (1.4 - 8.1 L/s/m2) range. The lower limit prevents channeling and the maximum rate allows higher service flows during those periods when other vessels are in regeneration. The maximum flowrate for mixed bed units in polishing service should not exceed 24 gpm/ft2 (16.3 L/s/m2).
3. Maximum and minimum bed depths should be 84 in. (2100 mm) and 30 in. (750 mm), respectively.
4. The maximum practical vessel diameter to be considered should be 15 ft (4.6 m).
5. Freeboard (as measured from the top of the exhausted resin bed) should be as follows:
Cation Units – 0.75 x resin bed depth + 42 in. (1050 mm)
Anion Units – 1.0 x resign bed depth + 48 in. (1200 mm)
Mixed Bed Units – 1.0 x resin bed depth + 48 in. (1200 mm)
6. Units should be designed with a maximum of one regeneration per 24 hours.
7. Forced draft degasifiers (also known as Decarbonators) typically reduce free carbon dioxide (CO2) to 5 ppm and oxygen (O2) to the 6 - 8 ppm range. The following design guidelines should be considered:
a. Maximum allowable water flowrate should be 17.5 gpm/ft2 (11.9 L/s/m2) of cross sectional tower area. Minimum tower height should be 16 ft (4800 mm).
b. The minimum allowable air flowrate should be 3 scfm per gpm (20 L/s per L/s) of design water flow.
c. Storage capacity should not be less than 10 minutes of design throughput rate. Additional storage capacity may be needed for regeneration water requirements.
Design of Ion-Exchange DM water plants is often vendor specific and manufacturers / vendors often provide performance guarantees for outlet DM water quality, regeneration chemicals consumption and efficiency in terms of cubic meter or gallon of Demineralized water produced per cubic meter or gallon of raw water feed to the DM water plant. However, the above mentioned guidelines can help process engineers in evaluating and making an informed judgement on the offers and specifications submitted by the vendors / manufacturers.
Hope readers of my blog find this blog entry informative and I look forward to comments from them.
Regards,
Ankur.

 FB
FB







Thank Ankur for your post containing very good recapitulated information. I would like to add in some points
1. H2SO4 is used with precaution to the Ca/Ba/Sr - SO4 precipitation at resin local or even in the regeneration conduct pipe
2. WAC/WAB are normally used to remove specific ions and preceded/followed by other strong resins types.