Edited by peterzwart, 12 June 2020 - 05:31 AM.
|
|
Posted 12 June 2020 - 05:25 AM

Edited by peterzwart, 12 June 2020 - 05:31 AM.
Posted 12 June 2020 - 08:44 AM
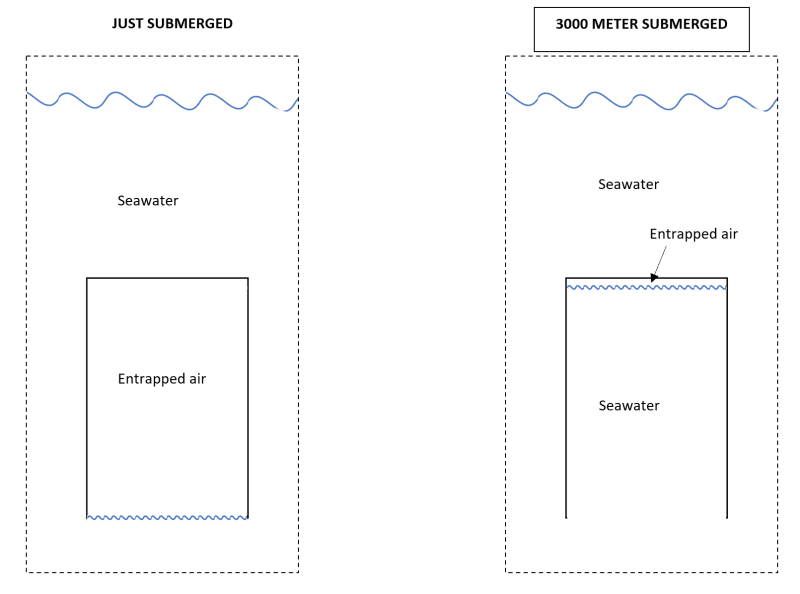
Air can dissolve into the water pretty quickly. I dont really have much rates to show, but I have experience with dissolving air into water at much lower pressures, <10bar.
I would think you can dissolve almost all the air at those kinds of pressures.
I normally refer to saturation curves to see how much air can dissolve in how much water. Unfortunately I don't have one that reaches the pressures you asked for.
Edited by thorium90, 12 June 2020 - 08:47 AM.
Posted 07 July 2020 - 04:56 AM
Thank you for your input Thorium90! Helpful and appreciated.
Peter
Posted 08 July 2020 - 10:04 AM
May be this one is helpful to you. we used this while calculating dissolved oxygen for material selection.
Calculating Gas Flow Rate Without CvStarted by Guest_worrier_* , 25 Nov 2025 |
|

|
||
Wet Scrubber Design For Low Flow Rate Of GasStarted by Guest_SIVAMOORTHY_* , 17 Apr 2018 |
|

|
||
Teg Dehydration Stripping Gas Flow RateStarted by Guest_SidRah_* , 20 Aug 2025 |
|

|
||
How To Estimate The Circulation Rate For Amdea (Mdea 45Wt + Pz 5Wt) BeStarted by Guest_tokotof_* , 29 Nov 2024 |
|

|
||
Psv Release Rate Vs TimeStarted by Guest_stu_* , 30 Nov 2024 |
|

|