Señor TALVI y demás miembros del Blog adjunto memorias de cálculos en Excel para su revisión técnica, ingeniería y conceptos que ustedes consideren que se deben tener en cuenta en este diseño.
La idea es que revisen estas memorias y la complementen con sus conocimientos y experiencia.
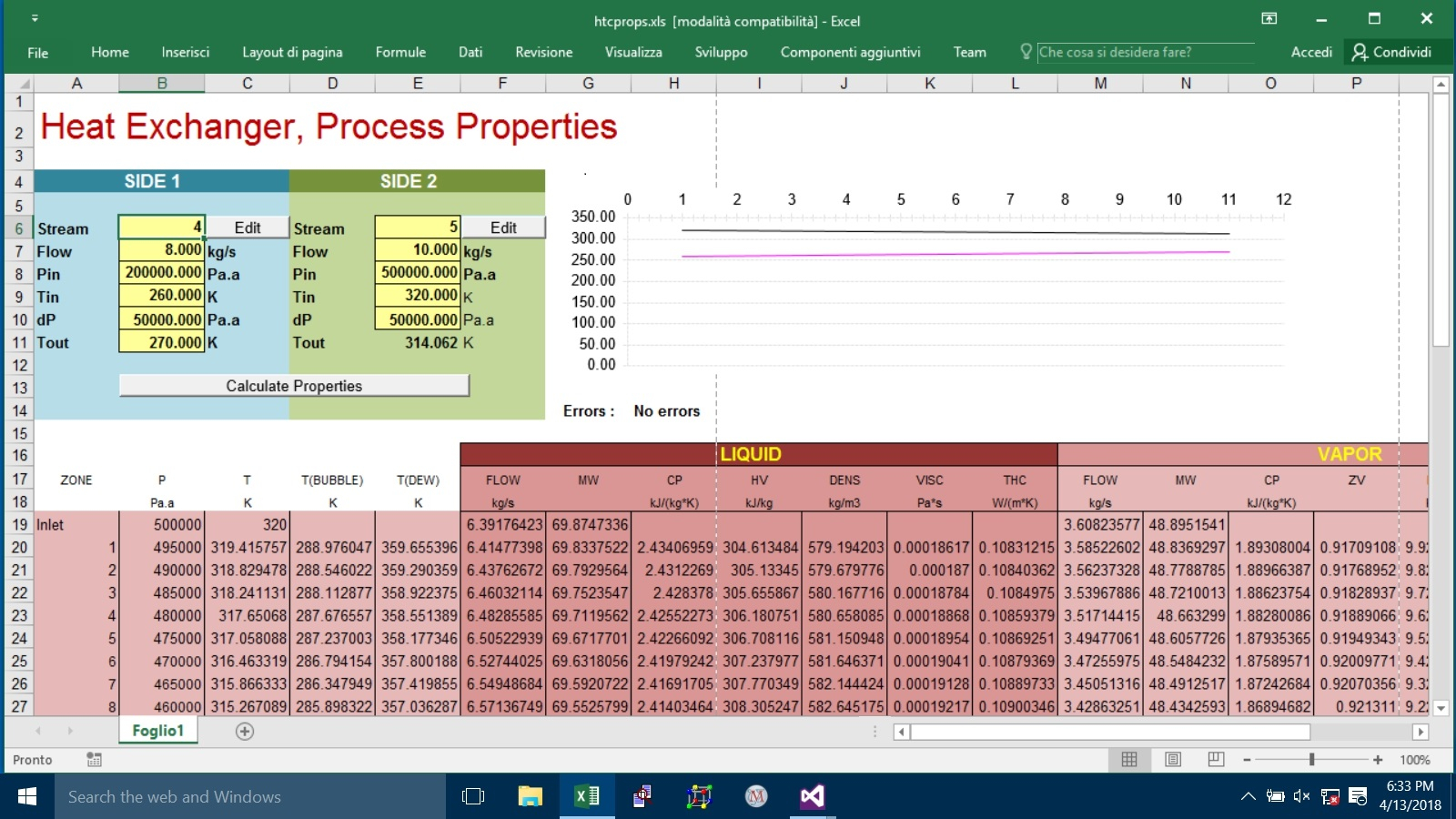
En este proyecto que estoy desarrollando se van a construir doce tanques así: Cuatro (4) de acero al carbono en SA-36, uno (1) en Acero inoxidable tipo 316 y seis (6) en PRFV o fibra de vidrio, con capacidad cada uno de 42.5 ton. para almacenar productos para químicos para tratamientos de aguas.
En las memorias están los datos del este producto del tanque N° 1 de acero al carbono. Mi objetivo es que este software en Excel le sirva a todos los miembros de la comunidad.
a la espera de su valioso aporte.
Saludos

 FB
FB
 Posted by
Posted by  Posted by
Posted by  Posted by
Posted by  Posted by
Posted by 

 Posted by
Posted by  Posted by
Posted by